|
Genereal Chemistry: Principles and Modern Applications 9th Ed. 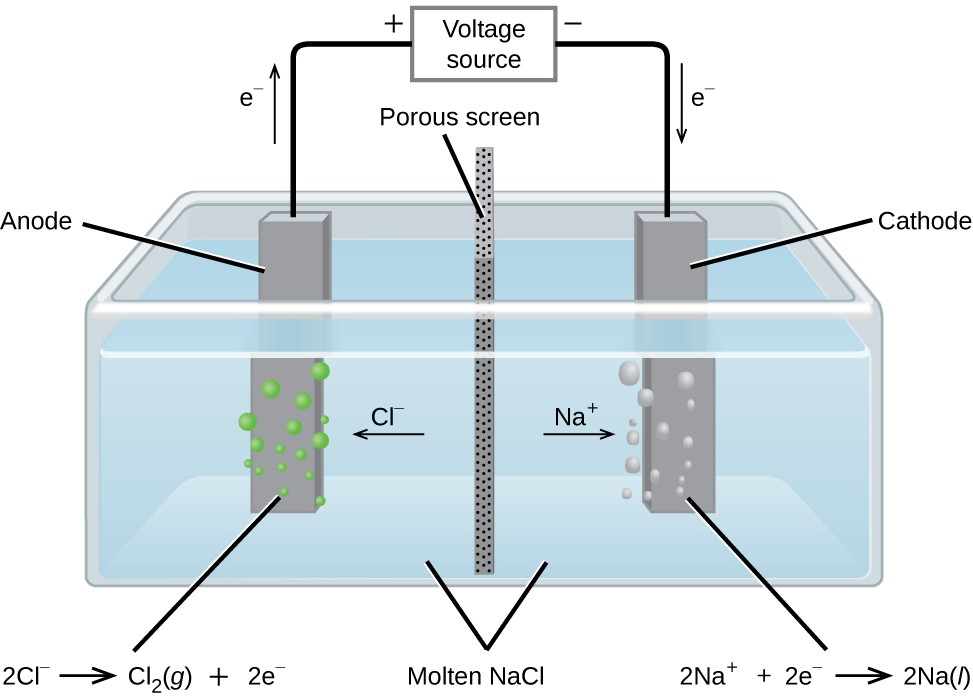
Write the equations for the anode reaction and the cathode reaction if the FeI2 is in aqueous solution (show work, including comparison with electrolysis of water). Write the equations for the anode reaction and the cathode reaction if the FeI2 is molten. Harwood, William, Herring, Geoffrey, Madura, Jeffry, and Petrucci, Ralph, General Chemistry: Principles and Modern Applications, Ninth Edition, Upper Saddle River,New Jersey, Pearson Prentice Hall, 2007. (4 pts.) Iron (II)iodide is electrolyzed.LiCoO 2 in lithium-ion batteries) will pull the electrons from the anode (e.g. “ Descriptive Principles of Chemistry”, C.V. When the circuit is closed, the stronger attraction for the electrons by the cathode (e.g. 
“Chemistry: The Central Science” Fifth Edition, Prentice-Hall, Inc. “Chemistry: The Central Science” Third Edition, Prentice-Hall, Inc. The terms were coined in 1834 by William Whewell, who derived the words from the Greek word Kathodes which means ‘descent’ or ‘way down’. “Chemistry: The Study of Matter and Its Changes”, John Wiley & Sons Inc 1993 Anode and cathode are defined by the flow of current. Thus, we reverse the other half-reaction to write it as an oxidation and the decomposition of sulfurous acid leads to the oxidation of sulfur dioxide to sulfate at the anode. The lack of concentrations indicates solutions are under standard conditions (i.e., 1 M) The silver reduction has a more positive E(red), so reduction is more spontaneous for 'Ag'+ and 'Ag'(+) is reduced at the cathode. Separate changes in phase by | and indicate the the salt bridge with ||. The anode always goes on the left and cathode on the right.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
